Links to prescribing information and adverse event reporting information can be found at the bottom of the page. For UK Healthcare professionals only.
Dosing, infusion, and administration
VECTIBIX®
DOSING AND ADMINISTRATION
Vectibix® treatment should be supervised by a physician experienced in the use of anti-cancer therapy. Evidence of wild-type RAS (KRAS and NRAS) status is required before initiating
treatment with Vectibix®. Mutational status should be determined by an experienced laboratory using validated test methods for detection of KRAS (exons 2, 3, and 4) and NRAS (exons 2, 3, and 4) mutations.
Before infusion
Flush the infusion line with sodium chloride solution to avoid mixing with other medicinal products or intravenous solutions.1
Vectibix® should be diluted in sodium chloride 9 mg/ml (0.9%) solution for injection by healthcare professional using aseptic technique. Do not shake or vigorously agitate the vial. Vectibix® should be inspected visually prior to administration. The solution should be colourless and may contain visible translucent-to-white, amorphous, proteinaceous particulates (which will be removed by
in-line filtration). Do not administer Vectibix® if its appearance is not as described above. Using only a 21-gauge or smaller diameter hypodermic needle, withdraw the necessary amount of Vectibix® for
a dose of 6 mg/kg. Do not use needle-free devices (e.g. vial adapters) to withdraw vial contents. Dilute in a total volume of 100 ml. The final concentration should not exceed 10 mg/ml. Doses higher than 1,000 mg should be
diluted in 150 ml sodium chloride 9 mg/ml (0.9%) solution for injection. The diluted solution should be mixed by gentle inversion, do not shake.
Discard the vial and any liquid remaining in the vial after the single-use.
Vectibix® must be administered using a low protein binding 0.2 or 0.22 micrometre in-line filter, through a peripheral line or indwelling catheter.
No incompatibilities have been observed between Vectibix® and sodium chloride 9 mg/mL (0.9%) solution for injection in polyvinyl chloride bags or polyolefin bags.
Infusion times
Recommended
infusion time1:
infusion time1:
~60minutes
If first infusion is tolerated, then administer subsequent infusions over1:
30 to 60
minutes
Infuse doses of > 1000 mg over1:
~90
minutes
- Do not administer Vectibix® as an intravenous push or bolus1
- A reduction in the rate of infusion may be necessary in cases of infusion-related reactions (see section on Vectibix® dose and infusion adjustment)1
Recommended dose for infusion
6 mg/kg of bodyweight given once every 2 weeks.1
Modification of the dose of Vectibix® may be necessary in cases of severe (≥ grade 3) dermatological reactions (see section on Vectibix® dose and infusion adjustments for further information).
After infusion
Infusion-related reactions (occurring within 24 hours of an infusion) were reported in 5% of patients treated with Vectibix®, of which 1% were severe (NCI-CTC grade 3 and grade 4). Hypersensitivity reactions
occurring > 24 hours-post infusion have also been reported including a fatal case of angioedema that occurred more than 24 hours after the infusion. Inform the patient of the possibility of late onset reactions and instruct
them to contact their physician if symptoms of a hypersensitivity reaction occur. Please refer to sections 4.4 and 4.8 of the Vectibix® Summary of Product Characteristics for further details.
VECTIBIX® DOSE AND INFUSION ADJUSTMENTS
Skin reactions and dose adjustments
Modification of the dose of Vectibix® may be necessary in cases of severe (≥ grade 3) dermatological reactions as follows:
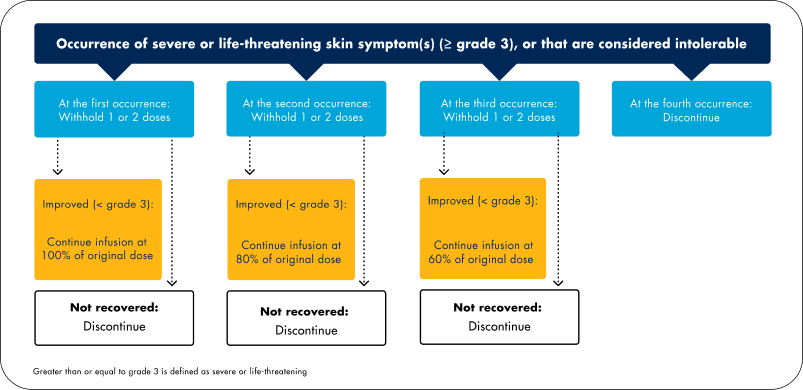
- Dermatologic-related reactions, a pharmacologic effect observed with epidermal growth factor receptor (EGFR) inhibitors, are experienced with nearly all patients (approximately 94%) treated with Vectibix®1
- Most skin reactions are mild to moderate in nature with 23% severe (grade 3) and < 1% life-threatening (grade 4); life-threatening and fatal infectious complications have been observed in patients who received Vectibix® monotherapy and in combination with chemotherapy1
- Pre-emptive versus responsive skin treatment reduced the incidence of grade ≥ 2 skin toxicities by ~50% in patients treated with Vectibix® in combination with chemotherapy2
- If your patient experiences dermatologic reactions that are grade 3 or higher, or that are considered intolerable, adjust the Vectibix® dose as recommended in the diagram above. Further details can be found in sections 4.2 and 4.4 of the Summary of Product Characteristics1
Infusion-related reactions and infusion rates
Infusion-related reactions (occurring within 24 hours of an infusion) were reported in approximately 5% of patients treated with Vectibix®, of which 1% were severe (grade 3 and grade 4); serious infusion-related
reactions have been reported, including rare post-marketing reports with a fatal outcome.1
Symptoms may include3:
- Headache
- Rashes
- Itching or hives
- Flushing
- Swelling (face, lips, mouth, around the eyes, and throat area)
- Rapid and irregular heartbeat
- Fast pulse
- Sweating
- Nausea
- Vomiting
- Dizziness
- Difficulty breathing or swallowing
- Decrease in blood pressure that may be severe or life-threatening and, very rarely, may lead to death
Hypersensitivity reactions occurring more than 24 hours after infusion have been reported, including a fatal case of angioedema that occurred more than 24 hours after the infusion.1
If a severe or life-threatening reaction occurs during an infusion or at any time post-infusion (e.g., presence of bronchospasm, angioedema, hypotension, need for parenteral treatment, or anaphylaxis) Vectibix® should be permanently discontinued.1
If your patient experiences an infusion-related reaction, follow the guidance below, as per the Summary of Product Characteristics.1

References
- Vectibix® Summary of Product Characteristics.
- Lacouture E et al. J Clin Oncol 2010;28:1351-1357.
- Vectibix® Patient Information Leaflet.
